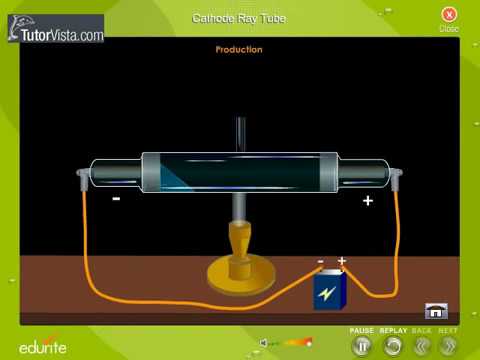
It was used for sending and measuring electric charges. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. Interactive: Build an AtomBuild an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change.J. These intermolecular forces prevent you from falling through an object like your chair. The electrons that surround all atoms are negatively charged and cause atoms to repel one another, preventing atoms from occupying the same space. Despite all this empty space, solid objects do not just pass through one another. Electrons have a mass of approximately 0 amu, orbit the nucleus, and have a charge of -1.Įxploring Electron PropertiesCompare the behavior of electrons to that of other charged particles to discover properties of electrons such as charge and mass.Īccounting for the sizes of protons, neutrons, and electrons, most of the volume of an atom-greater than 99 percent-is, in fact, empty space. However, protons have a charge of +1, and neutrons are uncharged. Protons, neutrons, and electronsBoth protons and neutrons have a mass of 1 amu and are found in the nucleus. In these atoms, the positive and negative charges cancel each other out, leading to an atom with no net charge. " In an uncharged, neutral atom, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. Scientists define these charges as "+1" and "-1. When considering atomic mass, it is customary to ignore the mass of any electrons and calculate the atom's mass based on the number of protons and neutrons alone.Įlectrons contribute greatly to the atom's charge, as each electron has a negative charge equal to the positive charge of a proton. Therefore, they do not contribute much to an element's overall atomic mass. Therefore, the number of neutrons in an atom contributes significantly to its mass, but not to its charge.Įlectrons are much smaller in mass than protons, weighing only 9.11 × 10 -28 grams, or about 1/1800 of an atomic mass unit.

Although similar in mass, protons are positively charged, while neutrons have no charge. Scientists define this amount of mass as one atomic mass unit (amu) or one Dalton. Protons and neutrons have approximately the same mass, about 1.67 × 10 -24 grams. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. Structure of an atomElements, such as helium, depicted here, are made up of atoms. This can be determined using the atomic number and the mass number of the element (see the concept on atomic numbers and mass numbers). The hydrogen atom (H) contains only one proton, one electron, and no neutrons. Atoms have different properties based on the arrangement and number of their basic particles.

The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Many biological processes are devoted to breaking down molecules into their component atoms so they can be reassembled into a more useful molecule.Ītoms consist of three basic particles: protons, electrons, and neutrons. For example, water is composed of hydrogen and oxygen atoms that have combined to form water molecules. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. It is equal in mass to a proton or it weighs 1 amu.Īn atom is the smallest unit of matter that retains all of the chemical properties of an element.